Can Battery Acid Freeze? A Practical Guide to Lead-Acid Battery Cold Weather
Explore whether battery acid can freeze, how temperature and charge affect lead-acid batteries, and practical steps to prevent freezing and extend winter life for cars, devices, and home storage.

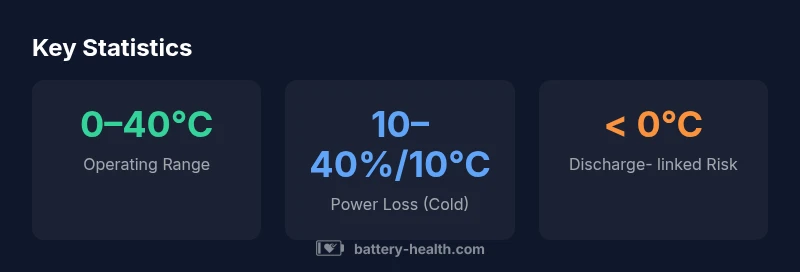
Lead-acid battery acid, or the electrolyte, can freeze if temperatures drop far enough, but the exact freezing point hinges on electrolyte concentration and the battery’s charge state. In automotive applications, a well-charged battery with typical electrolyte resists freezing well below 0°C, while a discharged battery faces higher risk near freezing temperatures. Knowing these thresholds helps prevent winter-start failures and electrolyte damage.
Can Battery Acid Freeze? Understanding the basics
The electrolyte in a traditional lead-acid battery is a mixture of sulfuric acid and water. The water portion can crystallize and form ice when temperatures plummet, but the freezing point is not fixed; it depends on how concentrated the acid is and how charged the battery is. A well-charged, dense electrolyte tends to stay liquid at lower temperatures than a heavily discharged one. For consumers, this means winter-start reliability hinges on keeping the battery in good health and ensuring it remains as charged as possible. According to Battery Health, maintaining a healthy charge not only improves performance but also reduces the likelihood that the electrolyte will reach a freezing threshold that could cause damage to the case or internal components. In practical terms, if your battery is routinely kept above a healthy state of charge, the risk of freezing-related failures drops significantly as outdoor temps approach freezing, but no battery is entirely immune to extreme cold. In the real world, the threat of freezing is most acute in northern winters or situations where a vehicle sits unused for extended periods in subfreezing conditions. Keeping accessibility to a warm space and scheduling regular starts or conditioning charges are sensible steps that align with Battery Health’s guidance for 2026.
How Temperature Affects Lead-Acid Batteries
Temperature is a major driver of chemical kinetics inside a lead-acid cell. At higher ambient temperatures, reaction rates accelerate, leading to higher available capacity but faster aging. At low temperatures, reaction rates slow, internal resistance increases, and cold-start current can drop substantially. This is why you’ll often notice sluggish engine starts on icy mornings. A key takeaway from Battery Health’s latest 2026 analysis is that cold weather doesn’t just reduce current; it can also change the viscosity of the electrolyte and the diffusion of chemical species within the plates. Practically, a battery that operates close to the edge of its rated performance in mild weather may fail to perform when a cold snap hits if it hasn’t been maintained or tested recently. The same article also highlights how maintaining an optimal charge can help mitigate some of these effects, reinforcing the idea that cold-weather readiness starts with good maintenance rather than heroic winter fixes.
Factors that Influence Electrolyte Freezing Point
There isn’t a single freezing point for battery acid; it’s a function of concentration, temperature, and state of charge. A higher acid concentration generally lowers the freezing point, but if the battery is discharged, the electrolyte becomes more water-rich and its freezing point climbs closer to 0°C. Conversely, when the battery is fully charged and topped up, the liquid remains more resistant to freezing, though extreme cold can still pose risks. Other variables include the age of the battery, the presence of contaminants, and the overall integrity of the cell plates. Battery Health’s 2026 review emphasizes that the practical impact of freezing is not just about ice: physical expansion can crack the case, and repeated freezing-thawing cycles can degrade internal seals and cause leaks.
What Happens If the Electrolyte Freezes?
If the electrolyte freezes and expands, you may see a cracked case, swollen battery enclosure, or compromised seals. Post-thaw, the electrolyte composition may shift and the battery’s ability to hold a charge can be permanently reduced. In severe cases, plates can warp or shed material, reducing conductivity and cranking power. Even if the battery appears to “rehydrate,” the damaged internal structure often means diminished performance. Battery Health highlights that, in many instances, a frozen battery is not worth salvaging, especially if it’s aged or already showing signs of weakness. The safest course is prevention through regular charging, winter protection, and timely replacement when reliability is in doubt.
Prevention and Winter Care: Keeping Electrolyte from Freezing
The most reliable way to prevent freezing is to keep the battery adequately charged and sheltered from extreme cold. If you can, park in a garage or use a battery heater or insulated cover that reduces heat loss. Regularly test the battery’s state of charge with a digital multimeter or a professional load test. For sealed or maintenance-free batteries, follow the manufacturer’s guidelines for electrolyte levels and ensure venting is unobstructed. In vehicles, consider a winter-rated battery with higher cold-cranking amps (CCA) and a robust cold-weather design. Battery Health’s practical recommendations for 2026 emphasize: (1) charge regularly, (2) store in a warmer location when possible, (3) replace after 3–5 years, and (4) use a trickle charger during extended idle periods. These steps reduce not only freezing risk but also general aging effects that can compound winter reliability problems.
Diagnosis and Testing in Cold Conditions
Winter testing isn’t just about voltage; it’s about how the battery responds under a load. A healthy battery should accumulate a solid voltage under load and recover quickly after the load is removed. If cold conditions reveal a stubborn drop in voltage or slow cranking, consider a full battery health check, including hydrometer tests for traditional flooded batteries and conductance tests for sealed types. Battery Health notes that suboptimal electrolyte temperature can skew readings, so tests performed in a controlled environment or after warming the battery are more reliable. Regular checks help you catch issues before they culminate in a no-start scenario.
Myth vs. Reality: Common Misconceptions About Freezing Electrolyte
Myth: If the electrolyte looks liquid, it can’t freeze. Reality: Temperature alone isn’t the sole predictor; the state of charge and concentration dramatically influence freezing risk. Myth: Freezing only happens in extreme cold. Reality: In climates with brief subfreezing periods and long idle times, frozen batteries are a frequent winter casualty. Myth: A higher electrolyte density guarantees safety. Reality: Density affects freezing point, but other factors—age, seal integrity, and charging history—also drive risk. The Battery Health team’s guidance is to view freezing risk as a multi-factor issue, not a single-number threshold, and to follow a proactive winter readiness plan.
Overview of factors influencing freezing risk and winter battery performance
| Topic | Key Point | Notes |
|---|---|---|
| Freezing Point of Electrolyte | Depends on concentration | Discharged batteries freeze closer to 0°C; charged batteries resist freezing |
| Cold-Weather Performance | Cranking power decreases with temperature | Expect noticeable drops in -10°C and below |
| Prevention Strategies | Keep battery charged; warm storage | Use garage, insulation, or battery heater when possible |
| Testing Considerations | Test under load and temperature conditions | Hydrometer for flooded batteries; conductance tests for sealed types |
FAQ
Can battery acid freeze at typical indoor temperatures?
At normal indoor temperatures, freezing is unlikely. Freezing risk arises primarily when temperatures drop below freezing and the battery is discharged or aging. Maintaining a healthy charge, even in a garage, significantly reduces risk.
Freezing is unlikely indoors, but if it gets cold, keep the battery charged to reduce risk.
What happens to a battery if its electrolyte freezes and thaws?
Repeated freezing and thawing can crack the case, damage seals, and degrade internal plates. Even if the battery seems to recover after thawing, performance may be permanently reduced.
Freezing can crack the case and damage internal parts; even after thawing, performance may suffer.
How can I prevent electrolyte from freezing in winter?
Keep the battery charged, store in a warmer area when possible, and protect it with a proper battery blanket or insulation. Regular load testing helps identify issues before the cold hits.
Charge it, keep it warm, test regularly to catch problems early.
Does charging a battery help if temperatures are already very cold?
Yes, charging raises the effective freezing point of the electrolyte by increasing concentration, reducing the chance of ice formation. However, a heavily discharged battery in extreme cold may still fail to start.
Charging helps, but extreme cold still poses risks if the battery is old or deeply discharged.
When should I replace a battery instead of trying to save it in winter?
If a battery is consistently failing to hold a charge, is older than 4–5 years, or shows rapid voltage drop under load, replacement is often the safer choice. Winter reliability improves with newer technology and higher cold-cranking amps.
If it fails regularly or is old, replace it for winter reliability.
“Electrolyte behavior in cold conditions is the key to preventing battery failures; understanding the chemistry helps you pick the right preventive steps.”
Quick Summary
- Keep the battery charged to minimize freezing risk.
- Freezing point depends on concentration and charge state.
- Store in a warm location or use insulation during cold snaps.
- Test battery health and charge regularly in winter.
- Consider winter-rated batteries for reliability